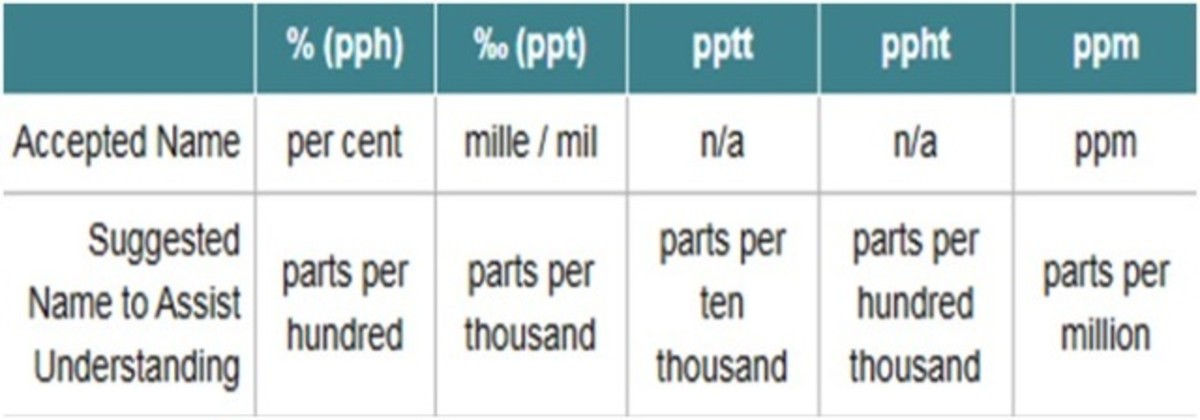
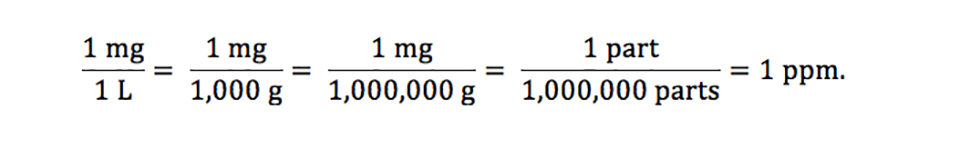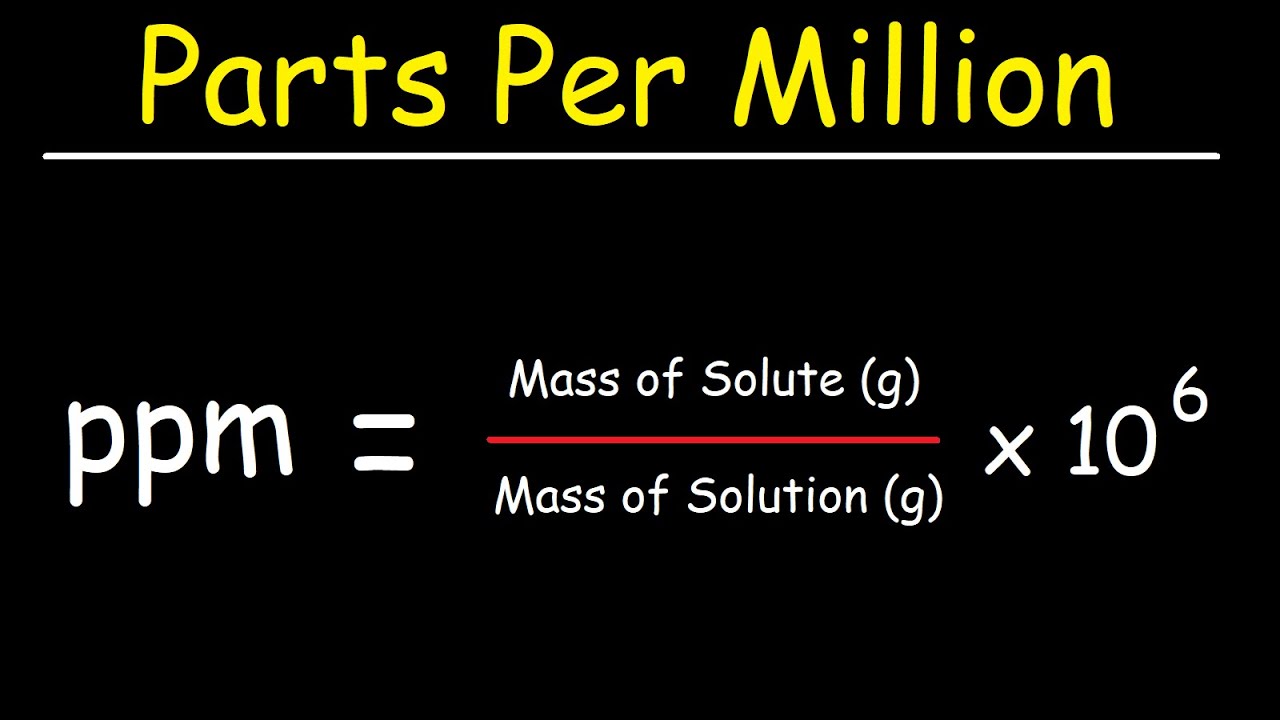10.30 mg of O2 is dissolved into a liter of sea water of density 1.03 g/mL . The concentration of O2 in ppm is .

Suppose a pure Si crystal has 5 × 10^28 atoms m^-3 . it is doped by 1 ppm concentration of pentavalent As. Calculate the number of electrons and holes. Given that n1 = 1.5 × 10^16m^-3 .

SOLVED: 77.14 pointt Zumchempa E 0I1 Hy Hotas Ask Your Toachar The units parts million (ppm) and parts per billion (Ppb) are commonly used enviranmental chemists In general; ppm mcans solutc ior

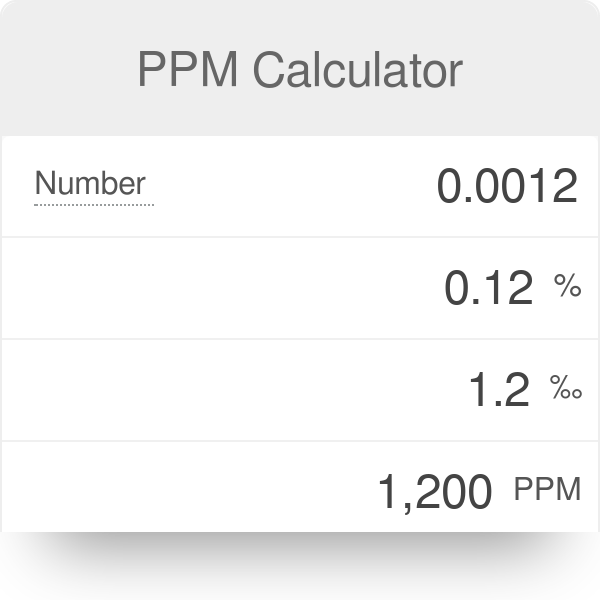
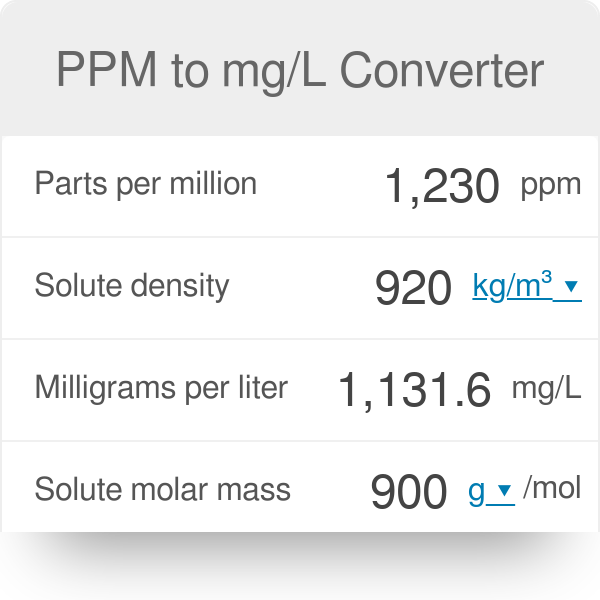
Units Conversion There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 *10-3 milligrams per liter (mg/l) are equal to parts per million (ppm) - ppt download