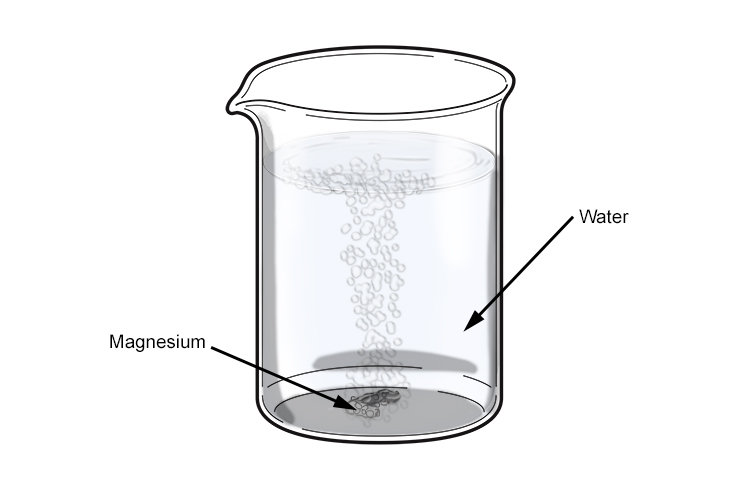
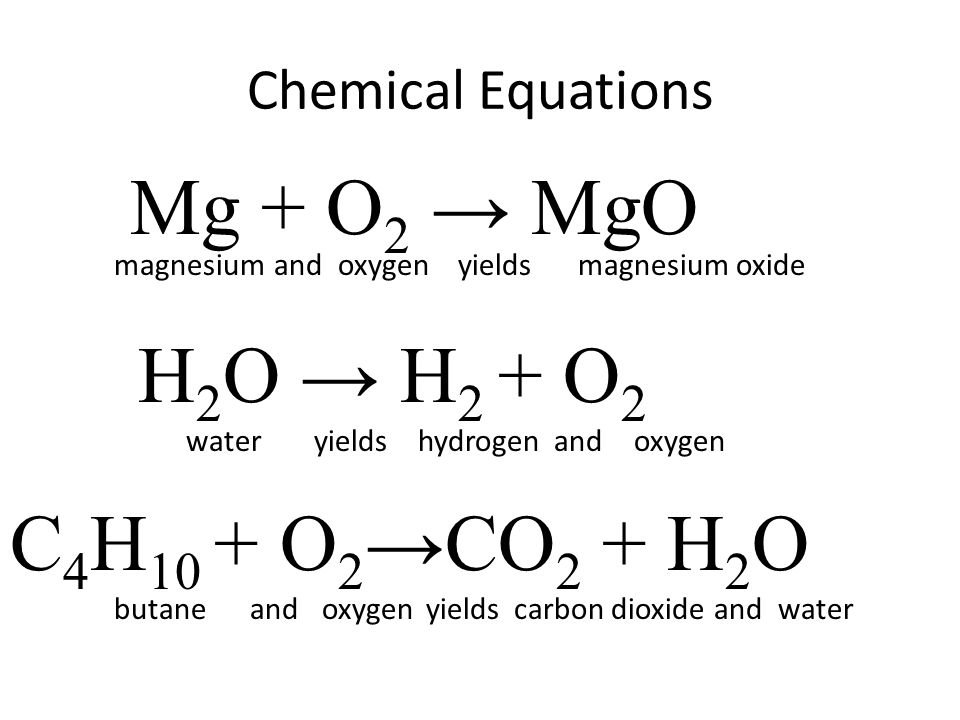
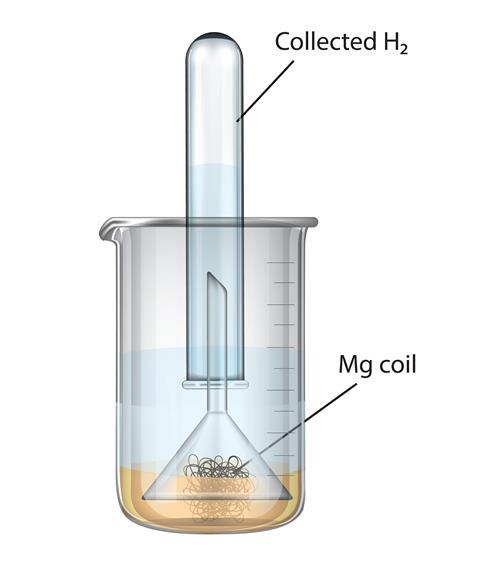
SOLVED: Magnesium metal reacts with water via the following balanced chemical equation: Mg (s) + 2 HzO Mg(OH)z (s) + Hz (g) If 4.73 g of magnesium is allowed to react with

Question Video: Identifying the Oxidized Specie in the Reaction of Magnesium Oxide with Hydrogen | Nagwa