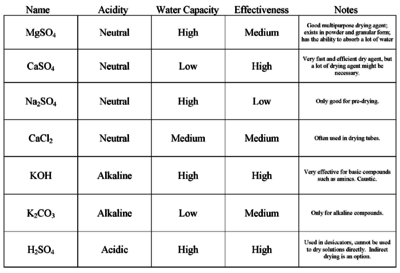
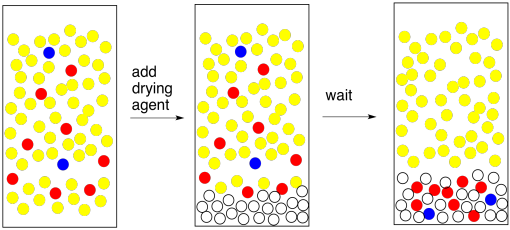
SOLVED: Upon separating the water (aqueous) layer from the hexanes (organic) layer, there will still be a small amount of water present in the organic layer, which will interfere with analysis and

Spectrum M1366-2.5KG (SPFDB) Magnesium Sulfate, Heptahydrate, FCC, MgSO4.7H2O: Amazon.com: Industrial & Scientific
![SOLVED: Sodium sulfate (NazSO4) and magnesium sulfate (MgSO4) are two drying agents which are [12%] commonly used in the lab; especially in extraction: A) Describe brielly; with chemical equations, how the drying SOLVED: Sodium sulfate (NazSO4) and magnesium sulfate (MgSO4) are two drying agents which are [12%] commonly used in the lab; especially in extraction: A) Describe brielly; with chemical equations, how the drying](https://cdn.numerade.com/ask_images/4ce331d8b187440a8339c1289cd1b7fa.jpg)
SOLVED: Sodium sulfate (NazSO4) and magnesium sulfate (MgSO4) are two drying agents which are [12%] commonly used in the lab; especially in extraction: A) Describe brielly; with chemical equations, how the drying