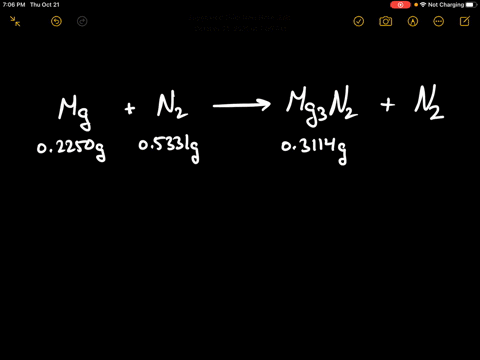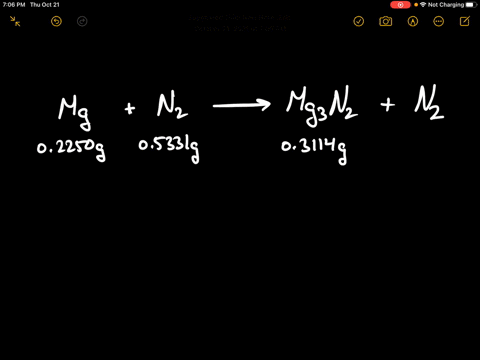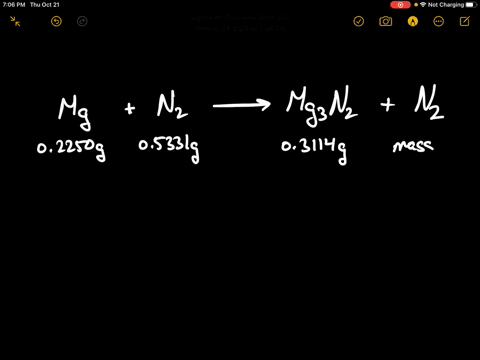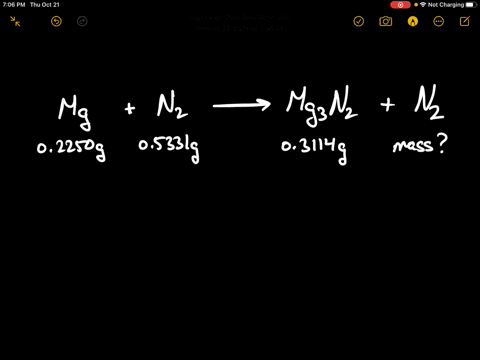
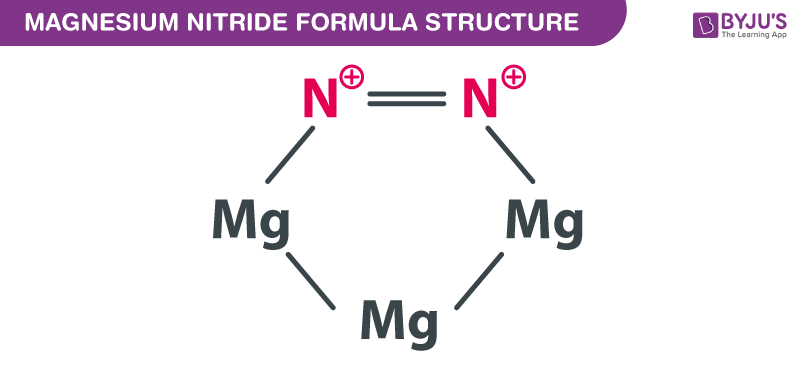
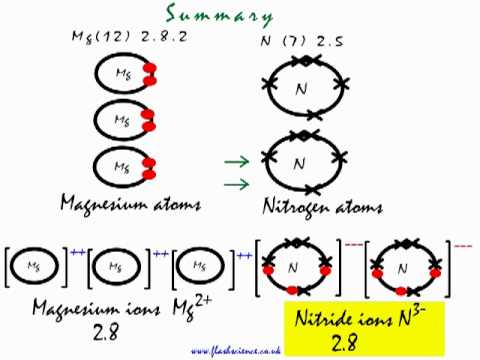
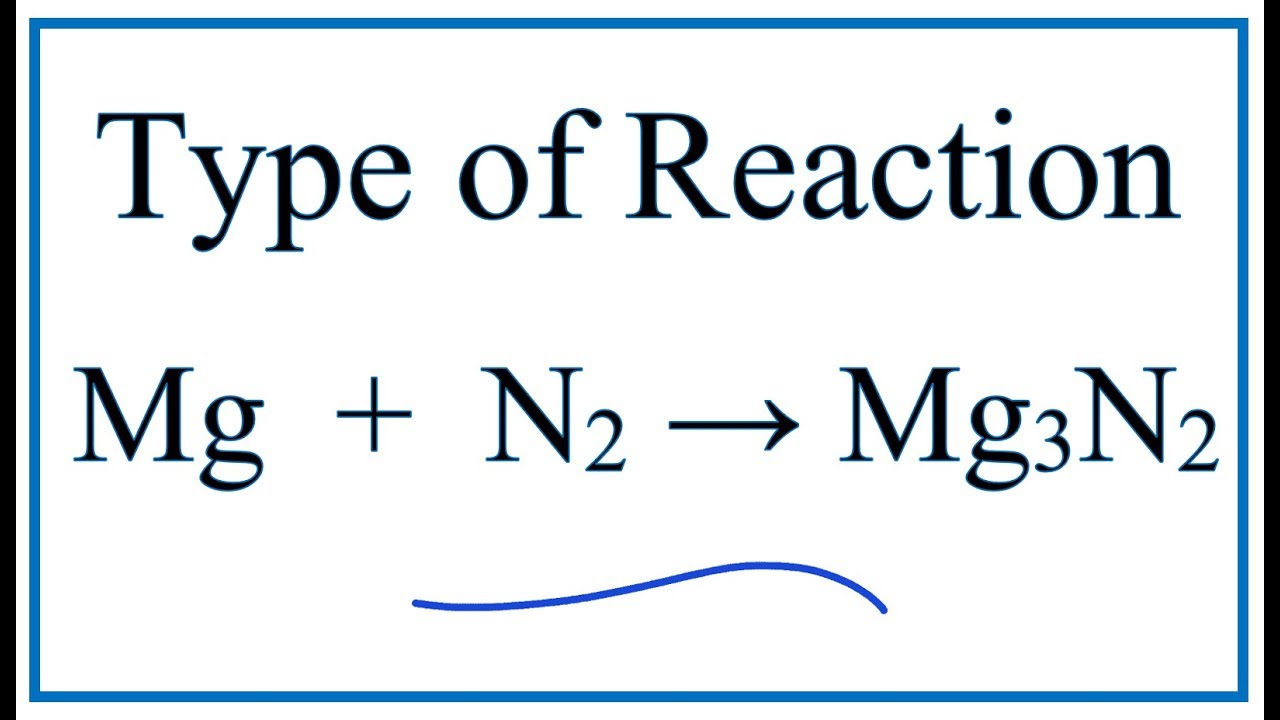Write the equations for the following and balance them Magnesium reacts with nitrogen to give magnesium nitride.

Indirect electrosynthesis of ammonia from nitrogen and water by a magnesium chloride cycle at atmospheric pressure - ScienceDirect

Starting with the atomic electron configurations of Mg and N to forming the corresponding | Homework.Study.com

Different levels of nitrogen and magnesium application on growth, yield... | Download Scientific Diagram

Do Now: What is a chemical reaction? What is a reactant? What is a product? Why do substances react with each other? - ppt download

Carbon monoxide reacts with hydrogen under certain condition to form methanol (CH3OH) . Write a balanced chemical equation for this reaction indicating the physical state of reactants and product, as well as
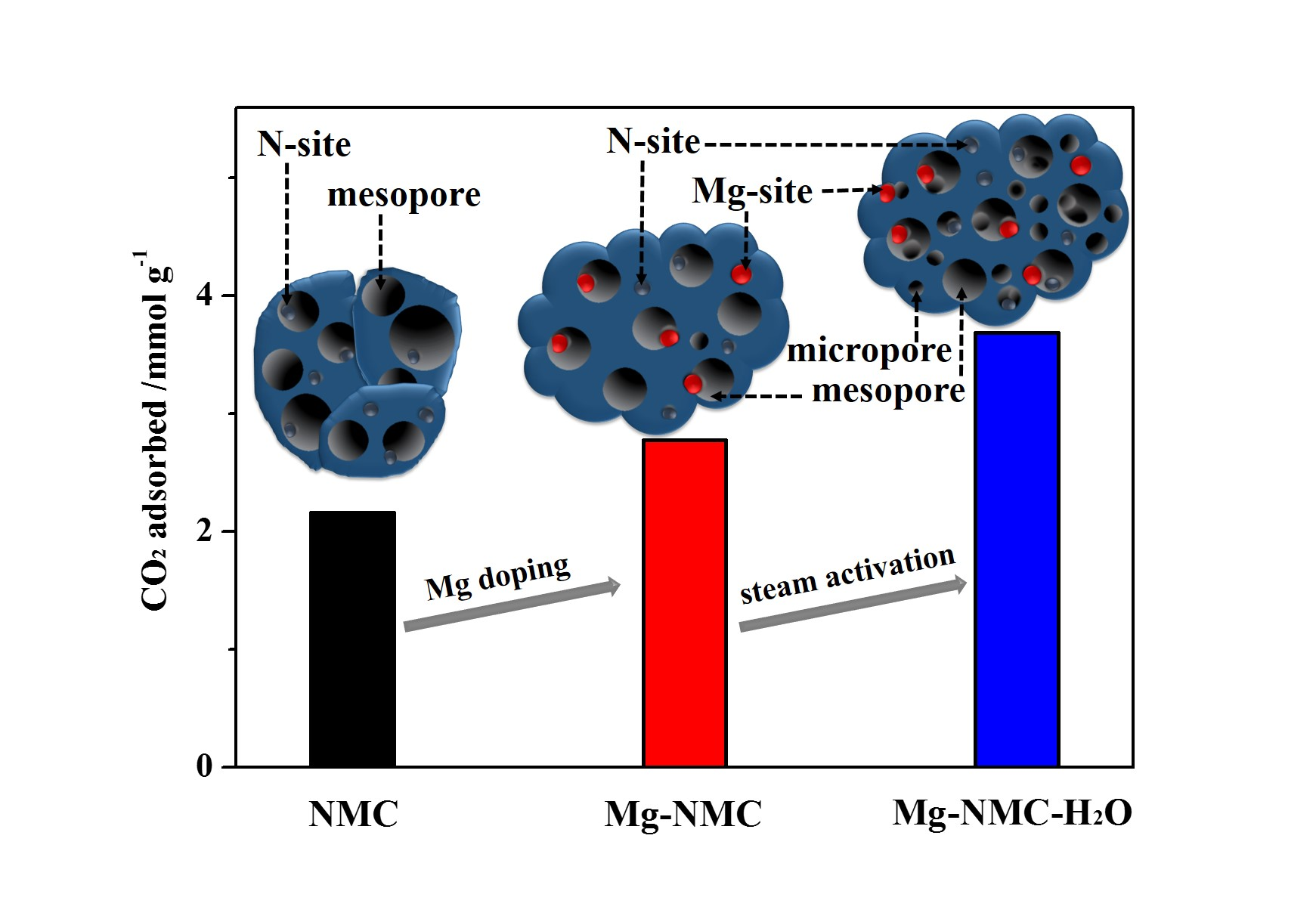
Nanomaterials | Free Full-Text | Magnesium and Nitrogen Co-Doped Mesoporous Carbon with Enhanced Microporosity for CO2 Adsorption

Mineral fertilizer - Magnesium Plus - Omex Agriculture - with trace elements / N / rich in magnesium