write chemical equation for reactions taking place when:-a) Manganese dioxide is heated with aluminium - Brainly.in

Had a go at making thermite. It was very difficult to ignite with magnesium. Any advice to improve the process? : r/chemistry

change the following reaction into balanced chemical equation a) magnese dioxide is heated with - Brainly.in

Minerals | Free Full-Text | Hydrometallurgical Production of Electrolytic Manganese Dioxide (EMD) from Furnace Fines

when a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and - YouTube
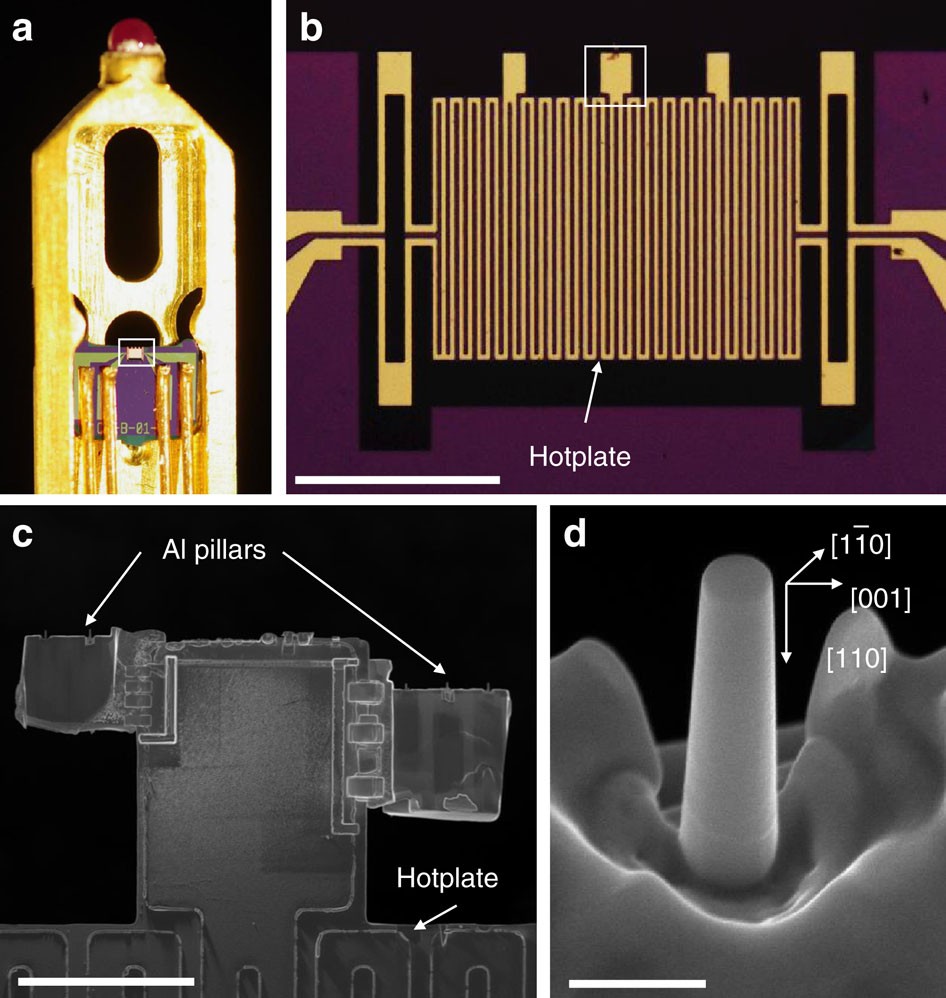
Effect of hydrogen on the integrity of aluminium–oxide interface at elevated temperatures | Nature Communications

The following reaction takes place when aluminium powder is heated with MnO(2) 3MnO(2)(s)+4Al(s)to3Mn(l)+2Al(2)O(3)(l)+"Heat" (a) Is aluminium gettuing reduced ? (b) zIs MnO(2) getting oxidised ?

a) Write the chemical reactions taking place when : (i) Manganese dioxide is heated with aluminium powder (ii) Steam is passed over red hot iron (c ) Magnesium reacts with hot water. (

Please answer question 7 and 8 5 6 7 8 10 What is the main - Science - Chemical Reactions and Equations - 12977479 | Meritnation.com

Write balanced chemical equation for Aluminium and manganese oxide - Science - Chemical Reactions and Equations - 13932711 | Meritnation.com
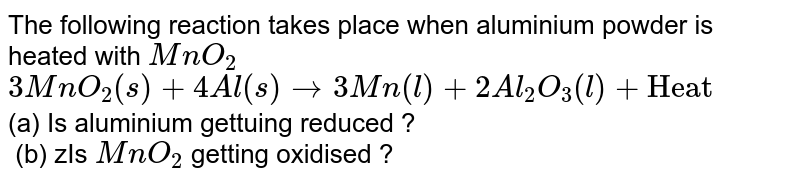
The following reaction takes place when aluminium powder is heated with MnO(2) 3MnO(2)(s)+4Al(s)to3Mn(l)+2Al(2)O(3)(l)+"Heat" (a) Is aluminium gettuing reduced ? (b) zIs MnO(2) getting oxidised ?