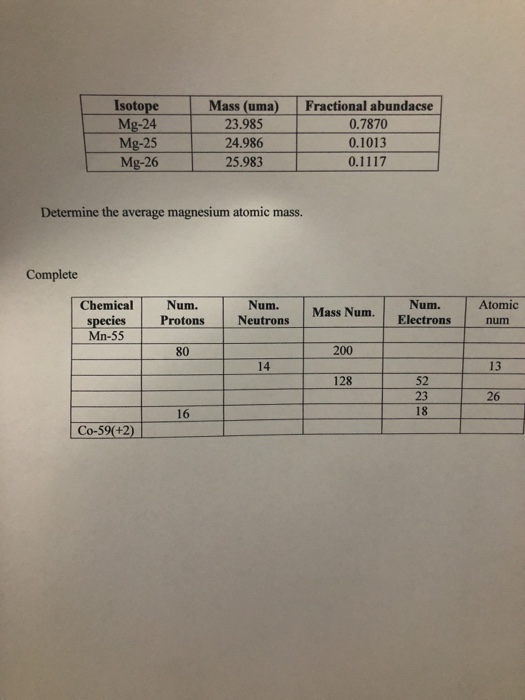
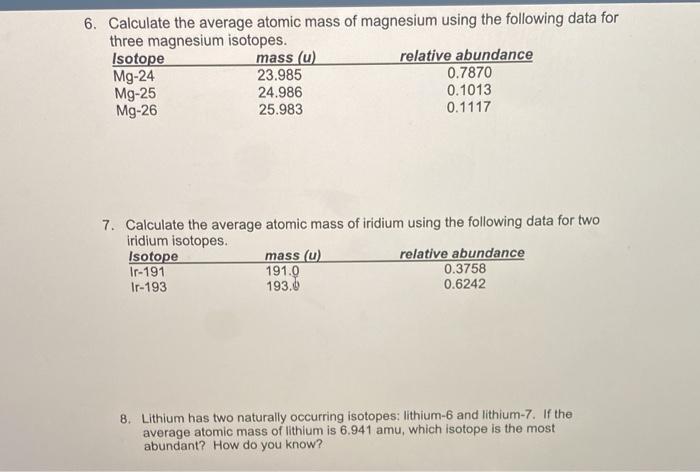
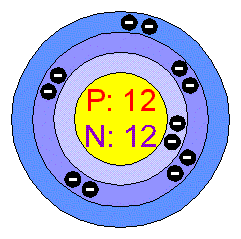
24^12Mg and 26^12Mg are symbols of two isotopes of magnesiumCompare the atoms of these isotopes with respect to: composition of their nuclei
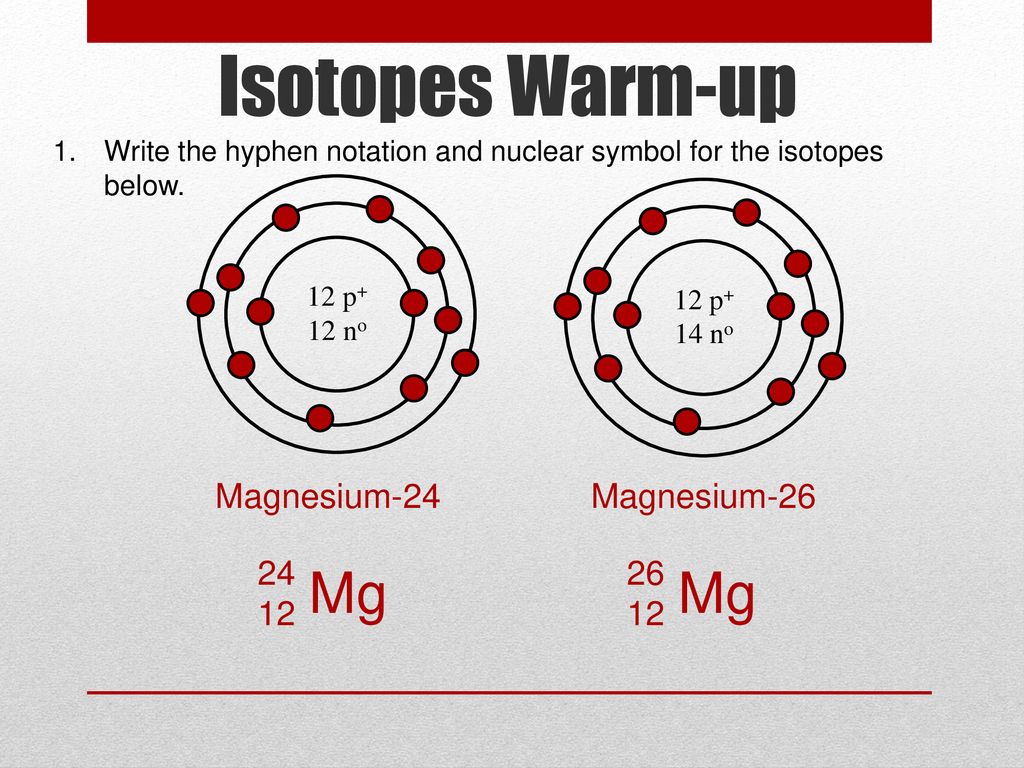
Isotopes Warm-up Write the hyphen notation and nuclear symbol for the isotopes below. 12 p+ 12 no 12 p+ 14 no 2. A atom has 26 protons and 30 neutrons. - ppt download
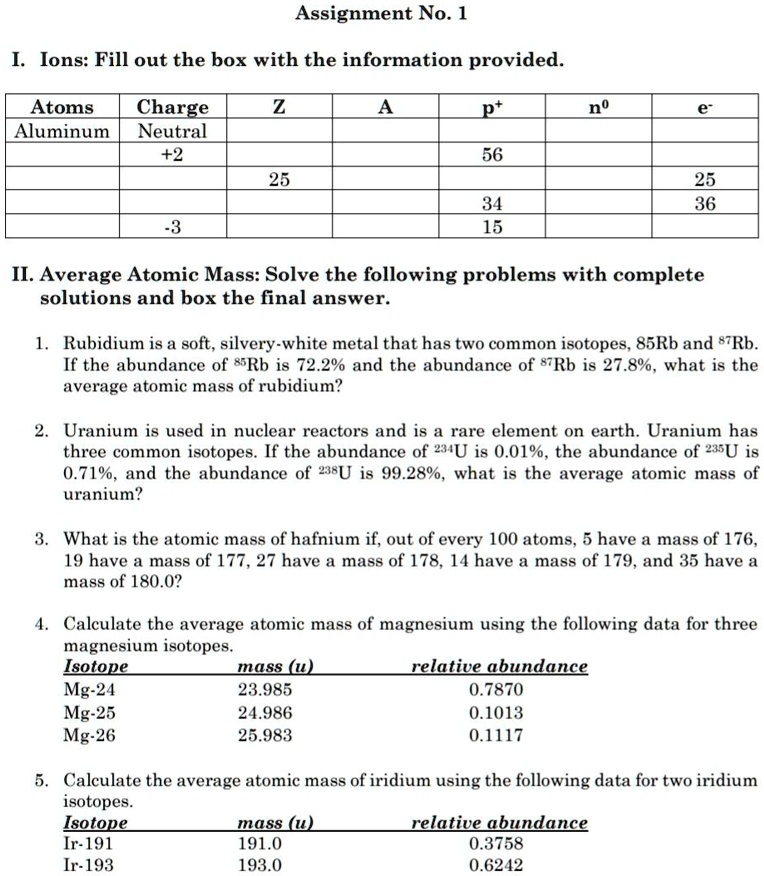
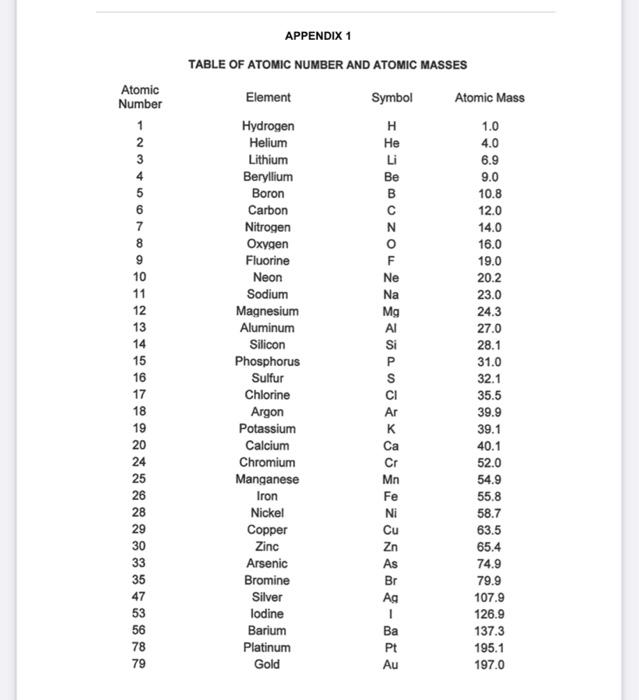
SOLVED: Assignment No. 1 Ions: Fill out the box with the information provided: Atoms Charge Aluminum Neutral +2 56 25 25 36 34 15 II Average Atomic Mass: Solve the following problems

Magnesium has two isotopes ^2412Mg and ^2612Mg . According to which isotopes of magnesium have different mass numbers.